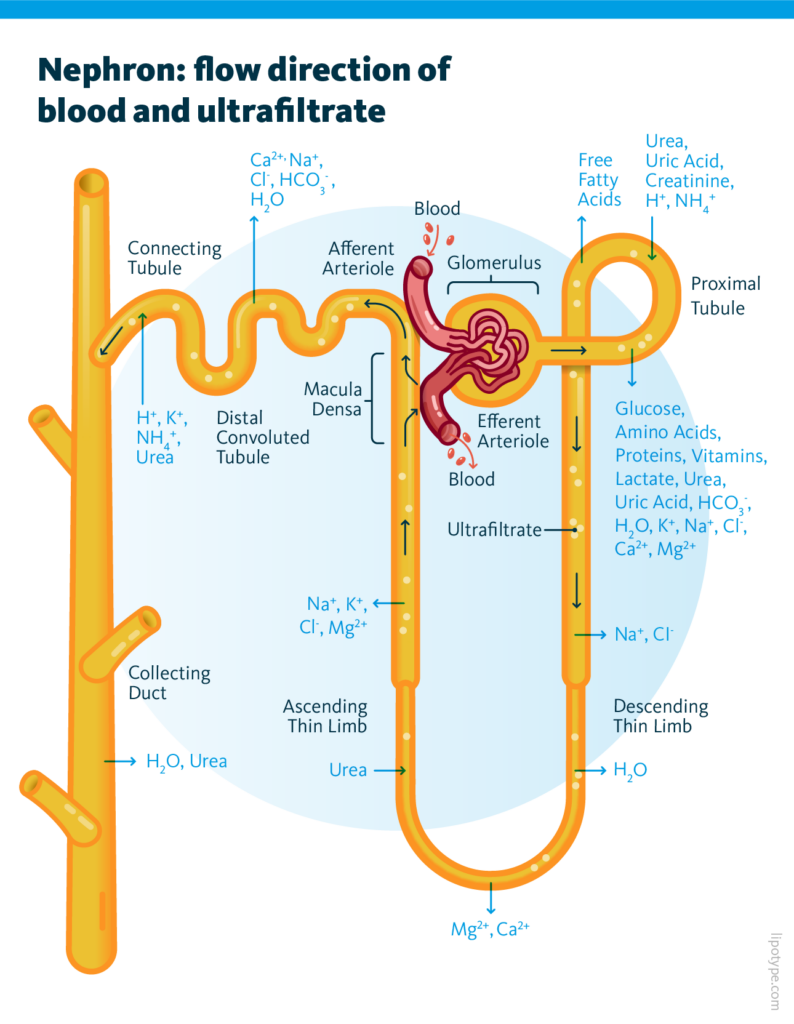
THE kidney is a paired organ that, among other functions, controls the water volume in the bloodstream by producing urine and removing waste and toxins. The kidney functional unit is called the nephron. The nephron’s work includes two steps: first, the glomerulus filters the blood producing ultrafiltrate, and then the tubule returns essential substances to the blood and removes wastes. During blood filtering, most of the solutes, water, glucose, and amino acids are reabsorbed in proximal tubule cells (PTC). This machinery requires energy, which mostly comes from fatty acids. The fatty acids are being stored in lipid droplets in segments 2 and 3 of PTC as well. The final product of these processes is urine.
During the course of diabetes, nephrons are damaged which negatively affects kidney function. One of the ways kidneys functional units can get damaged during diabetes is due to excessive accumulation of lipids leading to ‘lipotoxicity’ which is one of main drivers of kidney disease progression. Eventually a patient can develop kidney failure and would require artificial filtering (dialysis) and kidney transplant.
Perez-Marti and Simons studied lipid metabolism and the effect of accumulation of different saturated and monounsaturated free fatty acids in diabetic kidney PTCs. For this, they injected a low-dose streptozotocin (STZ) in a mouse model to destroy β-pancreatic islets and produce insulin deficiency leading to hyperglycemia and diabetes, and fed the mice with two types of high-fat diet enriched in either saturated fatty acids (SFA) or monounsaturated fatty acids (MUFA). In particular, the SFA-enriched diet contained butter with high amounts of palmitic acid and the MUFA-enriched diet contained olive oil with high amounts of oleic acid.
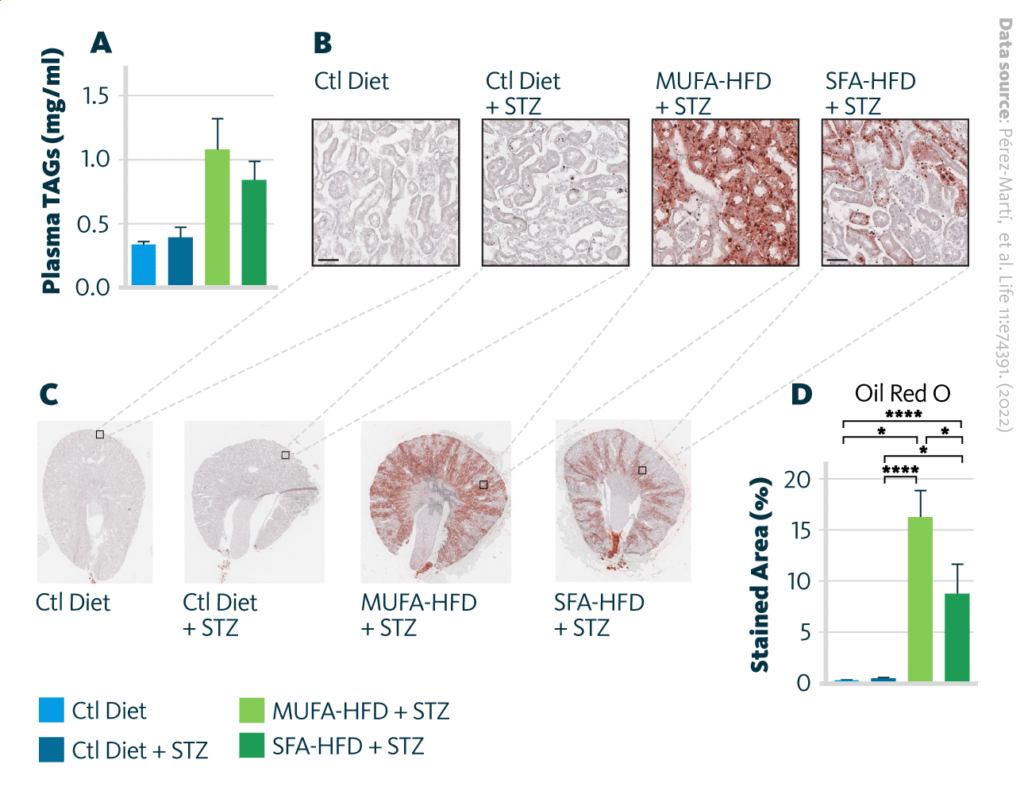
Accumulation of neutral lipids in kidney upon treatment with high-fat diet (SFA-HFD) and monounsaturated fatty acids high-fat diet (MUFA-HFD): A Plasmatic triacylglycerol (TAG) levels at week 16 after streptozotocin (STZ) injection increase upon HFD. B Representative bright-field images of whole kidney sections and C cortex magnification stained with Oil Red O (ORO). Scale bar: 50 μm. D Quantification of the stained cortex area reveals excessive lipid accumulation in kidney upon HFD. Data are presented as mean ± SEM; *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001; one-way ANOVA plus Holm–Sidak’s multiple comparisons test; n = 7 Ctl diet, n = 8 Ctl diet + STZ, n = 8 MUFA-HFD+ STZ, n = 7 SFA-HFD+ STZ.
Pérez-Martí, et al. Life 11:e74391. (2022) 10.7554/eLife.74391
The authors observed that a HFD enriched in saturated fatty acids caused tubular lipid droplet accumulation and fibrotic kidney damage in diabetic mice. The lipid droplet accumulation was assessed with microscopy of Oil Red O-stained samples, while the presence of fibrosis was visualized with Picro-Sirius Red. Interestingly, while MUFA are getting accumulated more than SFA in PTC, they cause substantially less fibrosis development.
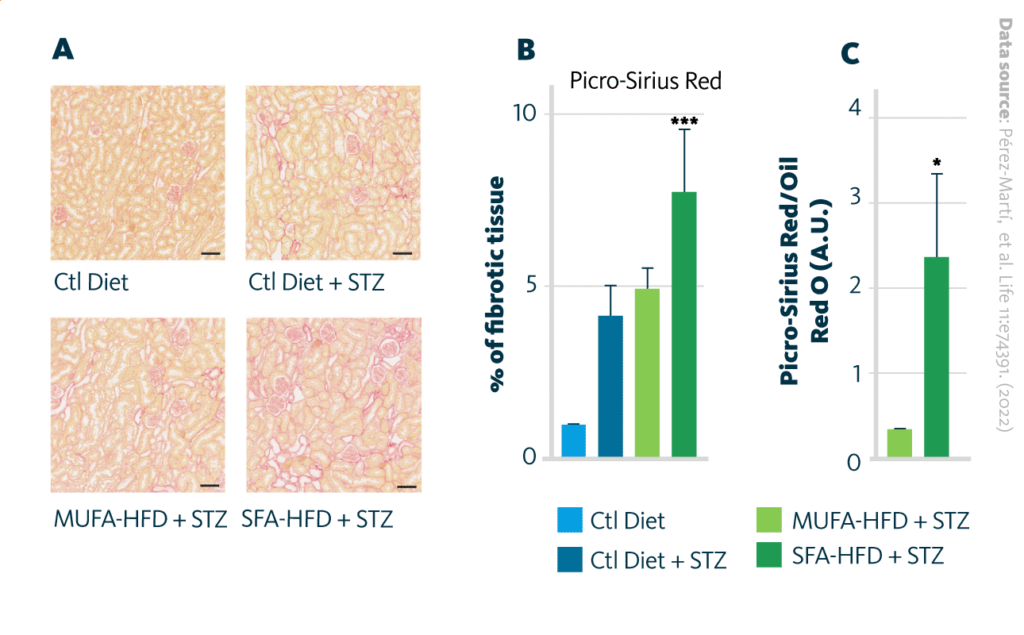
Induction of tubular damage by saturated fatty acids high-fat diet (SFA-HFD) and monounsaturated fatty acids high-fat diet (MUFA-HFD): A Bright-field images of kidney cortex stained with Picro-Sirius Red showing increased fibrosis development in all hyperglycemic mice with excessive fibrosis upon SFA-HFD. B Quantification of fibrotic tissue detected by Picro-Sirius Red and C normalized by fat deposition measured by ORO in mouse kidney cortex. Scale bar: 50 μm. Data are presented as mean ± SEM; *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001; one-way ANOVA plus Holm–Sidak’s multiple comparisons test; n = 7 Ctl diet, n = 8 Ctl diet + STZ, n = 8 MUFA-HFD+ STZ, n = 7 SFA-HFD+ STZ.
Pérez-Martí, et al. Life 11:e74391. (2022) 10.7554/eLife.74391
To better understand the molecular mechanisms driving diabetic SFA-mediated kidney damage as well as MUFA-mediated renoprotection, the scientists used proximal-tubule-like renal epithelial cells in their further cell culture experiments. Detailed analysis of this culture model showed not only changed lipid metabolism, but also revealed that palmitic acid impairs cell vitality, and this process can be rescued by adding of oleic acid to the cell culture media.
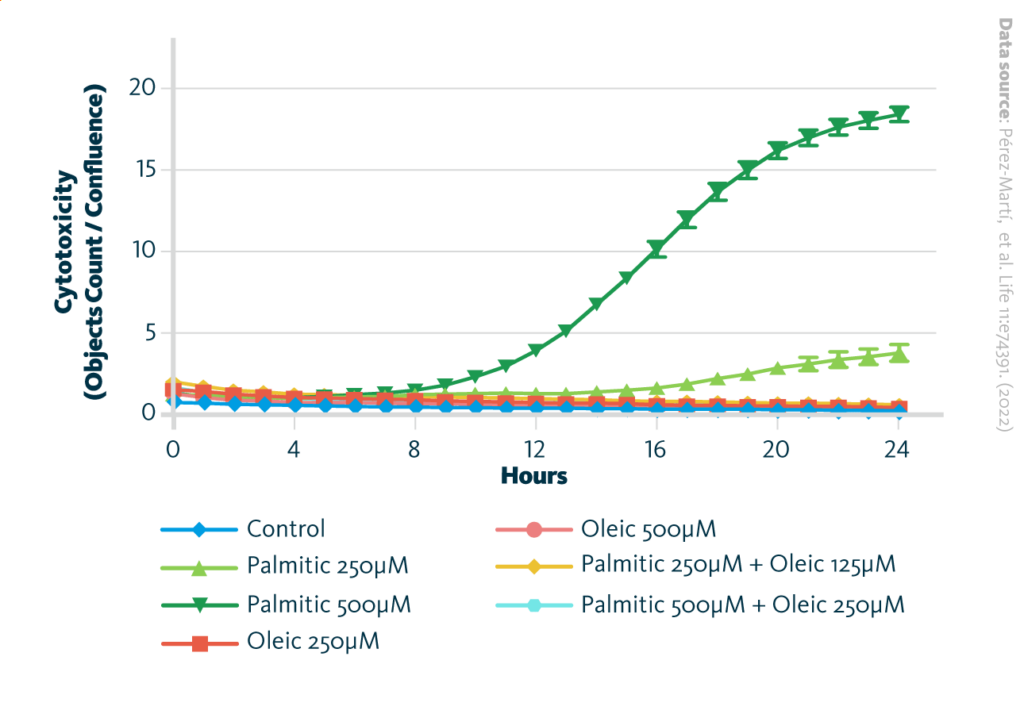
Palmitic acid-induced cytotoxicity is blocked by oleic acid in renal epithelial cells: Cytotoxicity throughout 24 hr incubation time in induced renal epithelial cells treated with several combinations of fatty acids. Data are presented as mean ± SEM; *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001; two-way ANOVA and Holm–Sidak’s multiple comparisons test; n = 3.
Pérez-Martí, et al. Life 11:e74391. (2022) 10.7554/eLife.74391
The researchers further investigated if oxidative stress response was involved in the effect of palmitic acid and oleic acid on cells and measured the generation of reactive oxygen species (ROS). Superoxide levels were increased by palmitic acid treatment, and when oleic acid was added together with palmitic acid, ROS levels returned to initial levels. Palmitic acid was found to increase mitochondrial activity while oleic acid reduced it. The combination of palmitic with oleic acids returned mitochondrial activity to the initial level as well, showing the antioxidant response.
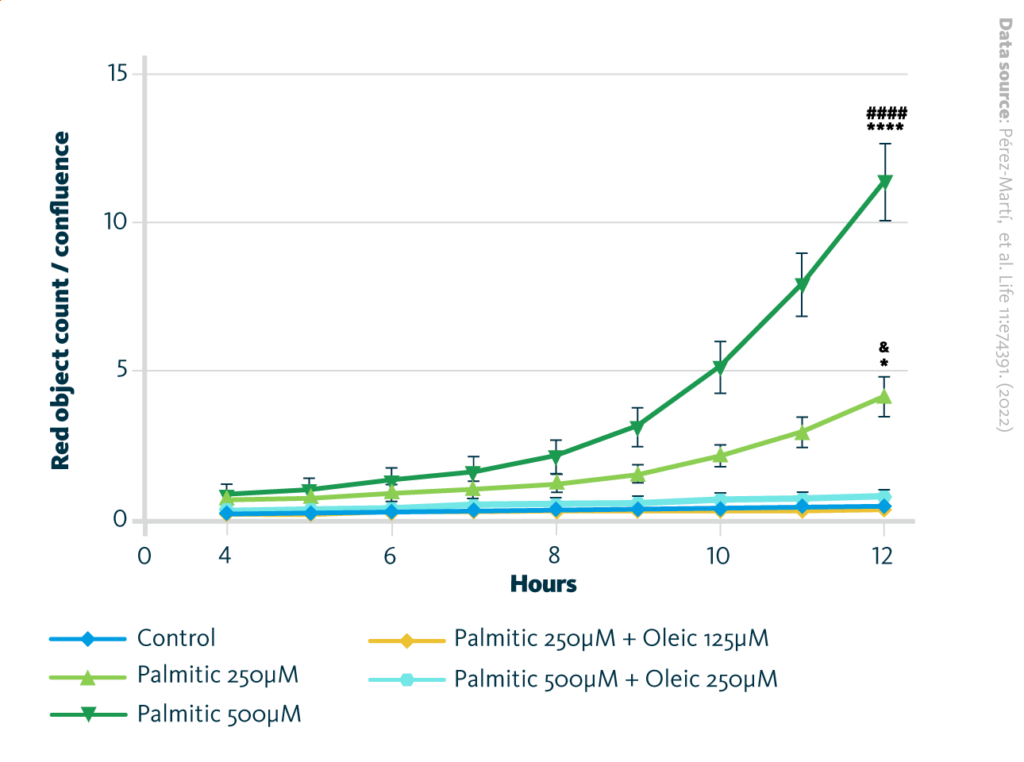
Quantification of reactive oxygen species (ROS) generation in renal epithelial cells treated with several fatty acids combinations: ROS generation in renal epithelial cells was visualized and further measured by incorporation of dihydroethidium (DHE) into the nucleus within 12h, and calculated as red object count to confluence. Data are presented as object count per well normalized by confluence, and as mean ± SEM; *p<0.05, ****p<0.0001 vs. control; ####p<0.0001 vs. palmitic 500 µM + oleic 250 µM; &p<0.05 vs. palmitic 250 µM + oleic 125 µM. One-way ANOVA and Holm–Sidak’s multiple comparisons test; n = 3.
Pérez-Martí, et al. Life 11:e74391. (2022) 10.7554/eLife.74391
The study further evaluates the role of endoplasmic reticulum stress (ER stress). ER stress can activate three different intracellular signal transduction pathways, collectively termed the unfolded protein response (UPR). The activation of the UPR leads to the expression of genes to restore ER homeostasis or trigger apoptosis if ER stress cannot be resolved. Activation of ER stress and UPR is also investigated in the development of vaccine adjuvants, as therapeutic target for triple negative breast cancer, and has been shown to occur in impaired microglia during remyelination.
In response to palmitic acid accumulation in renal epithelial cells, all ER stress response pathways are activated by palmitic acid after 16 hours of incubation. Again, this process was fully reversed by co-treatment with oleic acid. Further, the authors showed that the protective effects of oleic acid are specific for palmitic acid-induced ER lipid bilayer stress. Additionally, the effect of palmitic and oleic acid on ER membrane fluidity was measured: palmitic acid treatment decreased ER membrane fluidity, whereas oleic acid increased it.
To identify the lipids that might mediate palmitic acid-induced changes in ER membrane fluidity, renal epithelial cells treated with various fatty acids were subjected to shotgun lipidomics. In summary, oleic acid treatment led to the strong formation of triglycerides (TAG), and palmitic acid treatment led to increased levels of precursors of TAGs, such as diaglycerides (DAG), phosphatidic acid (PA), and lyso-phosphatidic acid (LPA).
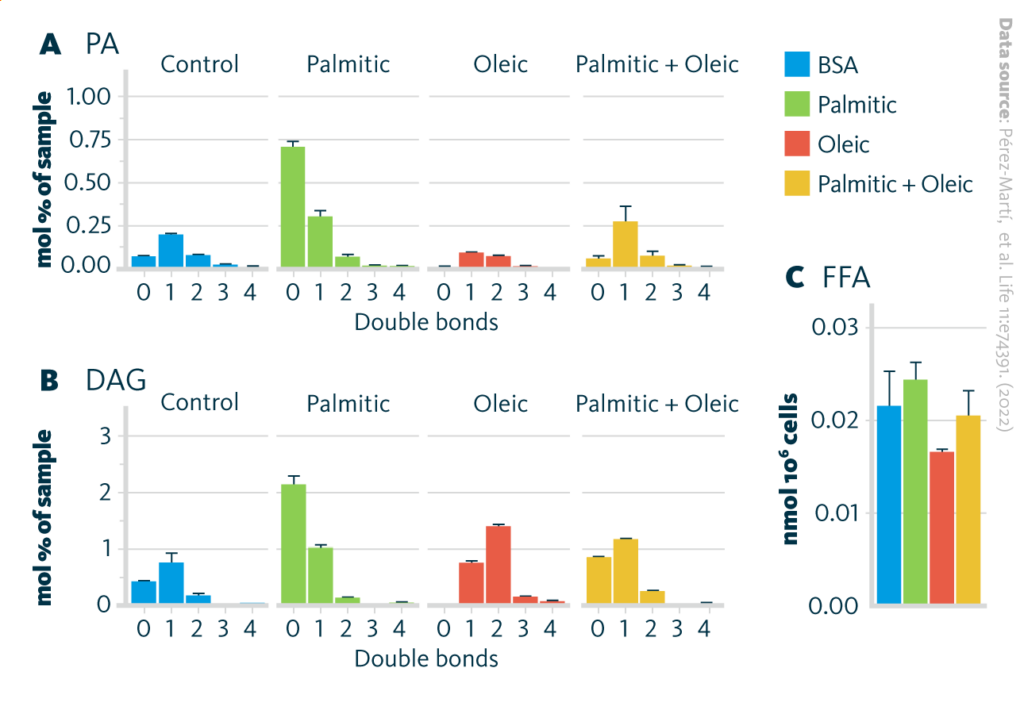
Lipidome of renal epithelial cells exposed to various fatty acids: A PA and B DAG species grouped by the number of double bonds in renal epithelial cells treated for palmitic, oleic, and palmitic + oleic acid. C Cytosolic-free fatty acids in renal epithelial cells treated for 16 hr with BSA, palmitic, oleic, and palmitic + oleic acid. Data are presented as mean ± SEM; one-way ANOVA and Holm–Sidak’s multiple comparisons test; n = 3.
Pérez-Martí, et al. Life 11:e74391. (2022) 10.7554/eLife.74391
The addition of oleic to palmitic acid enhanced the formation of TAGs, leading to reduced accumulation of DAG, PA, and LPA. Oleic acid also decreased the concentration of free fatty acids detected in cells, most likely due to stimulated TAG synthesis. Altogether, these results show that SFAs impair TAG production in cultured cells. Finally, a multiomics approach identified transcription factors potentially sensing ER membrane fluidity and driving adaptation mechanisms.
Further, the authors evaluated if increased TAG synthesis leads to the accumulation of lipids in lipid droplets and measured the number and volume of lipid droplets in renal epithelial cells under treatment with various fatty acids. It was shown, that MUFAs enhanced the formation of TAGs and, subsequentially, ER-derived lipid droplets.
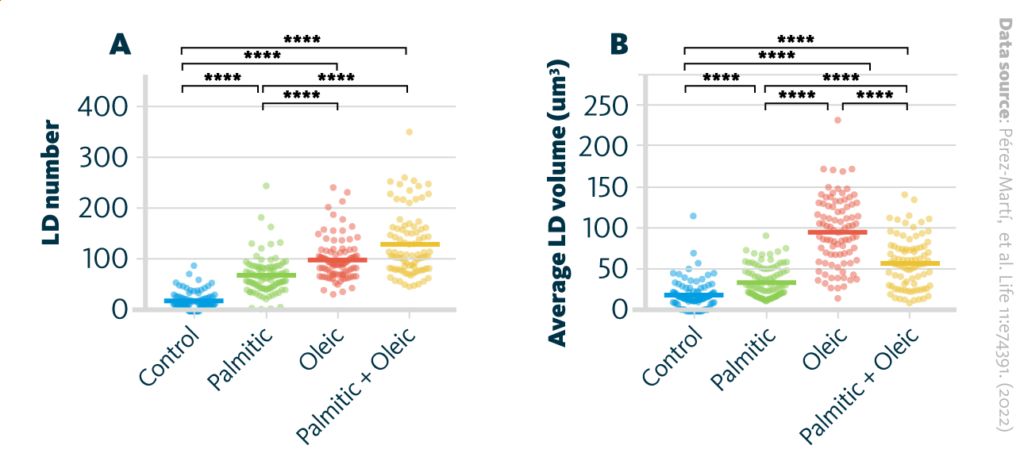
The formation of lipid droplets (LDs) upon treatment with various fatty acids: A LD number and B LD average volume in renal epithelial cells treated for 16 hr with palmitic, oleic, and palmitic + oleic acid. Data are presented as the mean + all values; *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001; Kruskal–Wallis plus Dunn’s multiple comparisons test; 10 cells per field from three fields were analyzed for three independent biological replicates.
Pérez-Martí, et al. Life 11:e74391. (2022) 10.7554/eLife.74391
Overall, the set of mice and cell culture experiments evaluated the disturbances in TAG synthesis as a key mechanism driving lipotoxicity in diabetic kidney disease. The diet in case of mice experiments, and enriched media in case of cell culture experiments, showed that oleic acid enhanced the production of TAGs while palmitic acid increased the saturation levels of the DAG species and impaired TAG synthesis. Taken together, these results support the protective role of TAG-enriched lipid droplet formation and provide insights into the mechanisms leading to lipotoxicity in diabetes.
Lipotype Lipidomics technology can be used to characterize the lipid profiles of cells and organoids. These data can shed light on metabolic diseases like diabetes and the subsequent damage they cause to particular organs and tissues.
Not sure how this works for your case?
Let’s talk it through!

Lipotype products are provided for Research Use Only. They are not intended for clinical diagnostic purposes and must not be used to inform medical treatment decisions. The content of this article is for scientific and educational purposes only and should not be considered medical advice.