What is the structure of Eicosanoids?
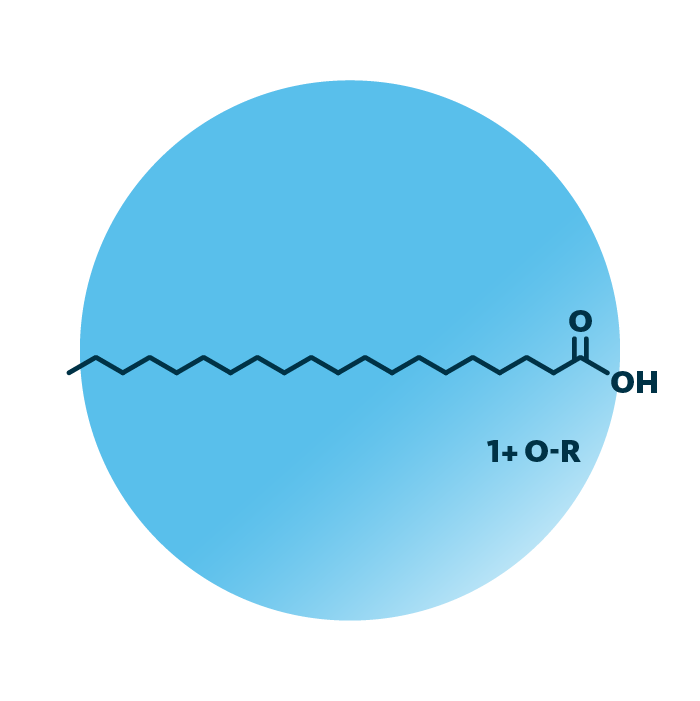
Eicosanoids belong to the fatty acyls. Their structure is based on eicosanoic acid, a twenty carbon atoms long hydrocarbon chain with a carboxyl group. Eicosanoids, docosanoids, octadecanoids, and further lipid groups are collectively termed oxylipins. Their hydrocarbon chain features oxygen-containing substituents, they are oxidized fatty acyls. Typically, eicosanoids contain multiple double bonds.
What is the structure of Eicosanoids?
Eicosanoids are found in eukaryotes such as animals, plants, and fungi, but rarely in prokaryotes. In cells, they are often in free form or linked to phospholipids to be released on demand. The composition of the hydrocarbon chain largely defines their biological function. Prostaglandins (PG), leukotrienes (LT), lipoxins (LX), and resolvin Es (RvE) are probably the most prominent eicosanoids. They are well-known for their capacity to modify inflammatory responses.
Thromboxanes (TX) are noted for their role in platelet aggregation. Isoprostanes (IsoP) are the ‘gold standard’ for non-invasive measurement of oxidative stress. Oxidized eicosadienoic (oxiEDE), eisocatrienoic (oxiETrE), eicosatetraenoic (oxiETE), and eicosapentaenoic acids (oxiPETE) serve diverse roles from being precursors to other eicosanoids and exerting signaling functions to modifying inflammation.