CELLULAR membranes are heterogeneous, flexible, 2D structures composed of lipids, proteins, and carbohydrates. They are the site of many essential biological functions, including signal transduction, nutrient/waste transport, and energy generation. To optimize membranes for each of these varied functions, cells tune membrane biophysical properties, such as structure, composition, and fluidity.
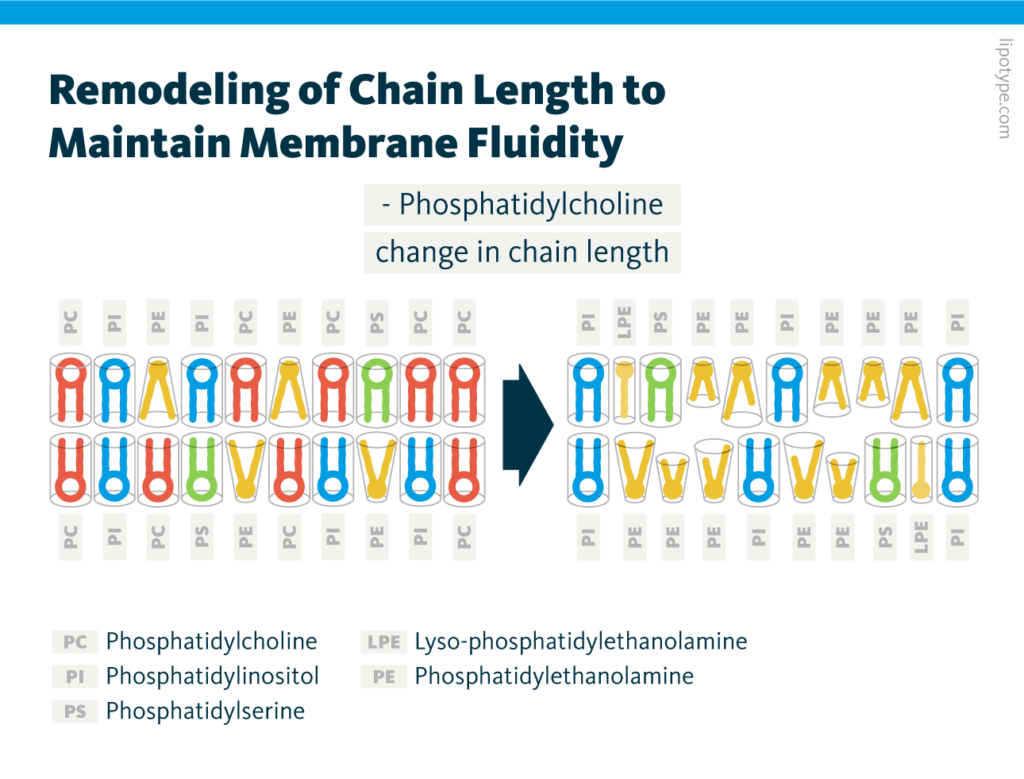
In eukaryotes, the glycerophospholipid phosphatidylcholine (PC) is abundant in the membrane – it can account for over 50% of membrane lipids – and has functional roles in signal transduction and metabolism. Changes in PC acyl chain composition are also linked to disease, possibly through changes in membrane fluidity, but this relationship has not been fully characterized.
Yeast is often used as a model eukaryote to study lipid function because it is tolerant to variations in lipid composition. A team of researchers from the Netherlands examined the function of PC in yeast. Their results identify changes in the organism’s morphology, gene expression, lipid composition, and membrane fluidity in the absence of PC.

Membrane phospholipids: Comparison of the general molecular structure of the ester phospholipid classes phosphatidylcholine, phosphatidylethanolamine, phosphatidylinositol, and phosphatidylserine, which are commonly found in cellular membranes.
To isolate yeast strains lacking PC, a yeast clone (cho2opi3) that requires external choline to synthesize PC was incubated in choline-free media. The resulting yeast lacked PC, exhibited changes in ploidy (co S(2n-1)) or a point mutation in the gene coding for the enzyme Acc1, acetyl-CoA carboxylase 1 (co acc1N/H), and displayed altered morphology.
To probe how yeast adapt to the lack of PC, whole-cell lipids were isolated and characterized via mass spectrometry-based shotgun lipidomics and thin layer chromatography. The PC-free yeast demonstrated an increase in total membrane lipid content and triacylglycerol concentration compared to wild-type and cho2opi3 yeast. In the absence of PC, phosphatidylethanolamine (PE) and phosphatidylinositol (PI) were the predominant components of the membrane.
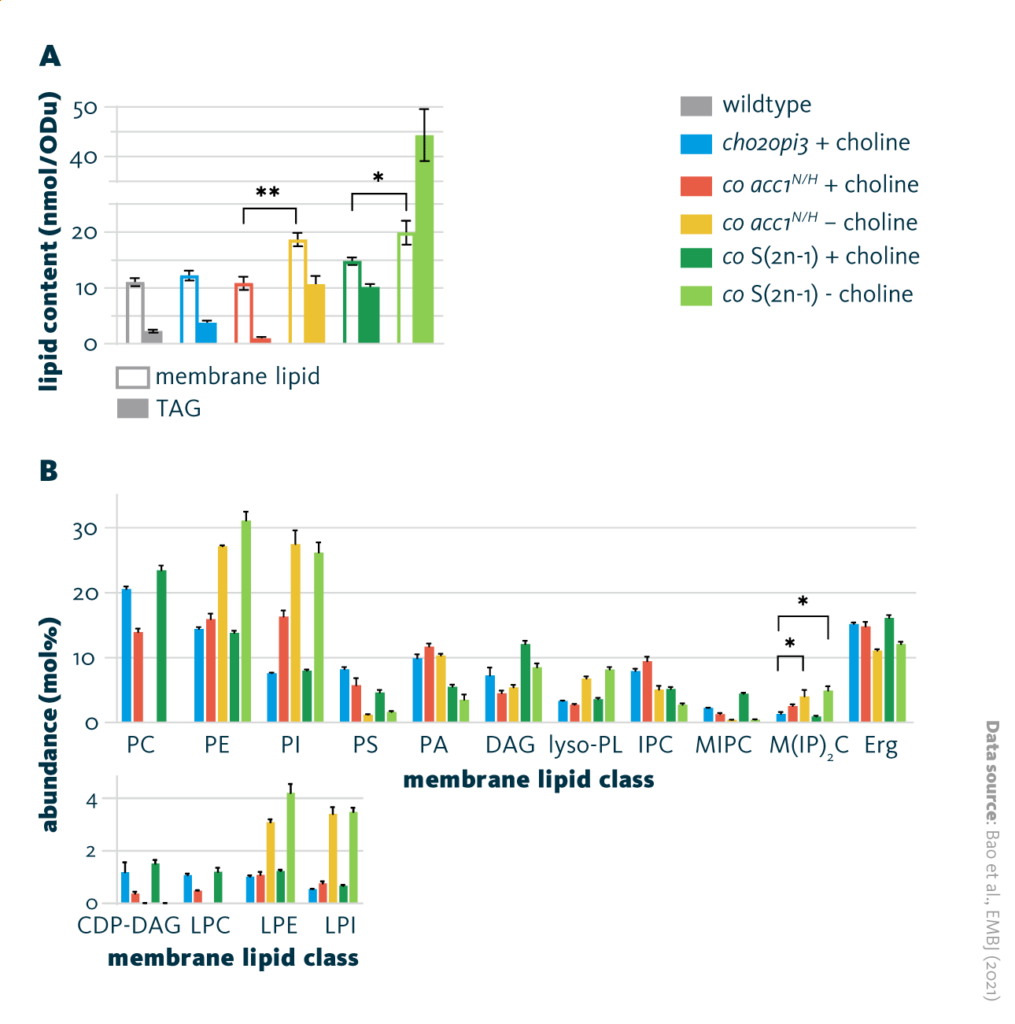
Lipidome remodeling of cho2opi3: Comparison of the lipid composition of different cho2opi3 suppressor yeast strains cultured with (+ choline) or without (- choline) 1mM choline. A Membrane lipid and triacylglycerol (TAG) content per OD600 unit, and B membrane lipid class composition.
Bao et al., EMBJ (2021), doi: 10.15252/embj.2021107966
Additionally, lipidomics analysis of yeast strains identified a marked reduction in acyl chain length in PC-free yeast compared to cho2opi3 yeast. Specifically, the abundance of 18-carbon fatty acids fell while that of 16-carbon and shorter fatty acids rose. This trend was observed for both triacylglycerols and membrane lipids. The saturation of phosphatidylethanolamine also increased.
Acc1 catalyzes fatty acid synthesis and can regulate chain length. To test the hypothesis that the Acc1 mutation in PC-free yeast reduces the enzyme’s activity, cho2opi3 yeast were treated with a strong Acc1 inhibitor. The yeast grew in the absence of choline and had shorter acyl chains, replicating the phenotype of PC-free yeast carrying an Acc1 mutation and supporting the hypothesis.
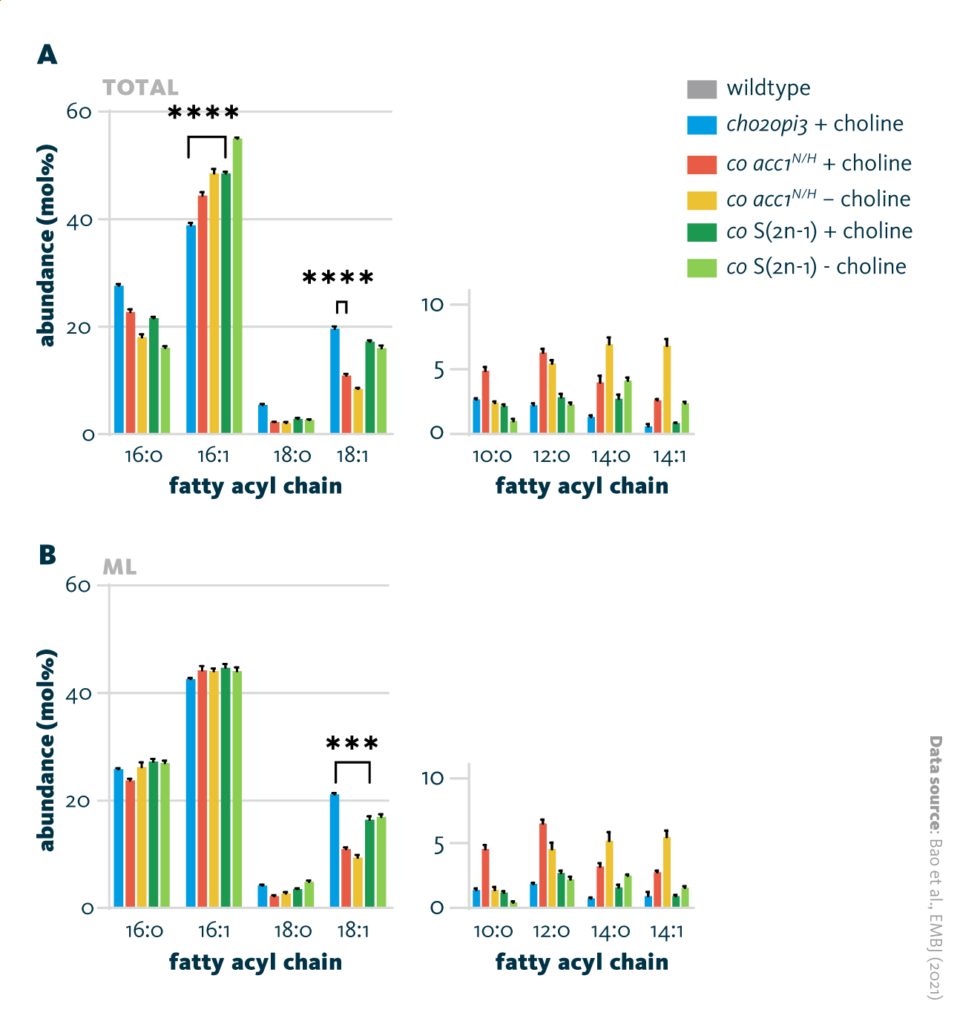
Fatty acyl chain remodeling of cho2opi3: Comparison of the fatty acyl chain of different cho2opi3 suppressor yeast strains cultured with (+ choline) or without (- choline) 1mM choline. A Fatty acyl profiles of the total lipid fraction, and B fatty acyl profiles of the membrane glycerolipids and phospholipids fraction.
Bao et al., EMBJ (2021), doi: 10.15252/embj.2021107966
A subsequent series of experiments demonstrated that PC-free yeast with changes in ploidy adapt to the lack of PC by increasing intracellular free fatty acid concentration, which upregulates expression of acyl-CoA, which in turn inhibits Acc1.

Membrane lipid packing: Comparison of intensity encoded GP-images of yeast strains with (+) and without (-) choline. White arrows point to plasma membranes exhibiting higher lipid packing than internal, organellar membranes, blue arrows indicate very highly packed lipids. White bars correspond to 10 µm.
Bao et al., EMBJ (2021), doi: 10.15252/embj.2021107966
An optical fluorescent reporter showed that changes in lipid composition alter membrane fluidity in these yeast strains. Specifically, cho2opi3 yeast had lower fluidity, likely due to high PE content. However, membrane fluidity was restored to wild-type levels in PC-free yeast, which is attributed to the shortened acyl chains.
Overall, this study demonstrates that yeast can adapt to the absence of PC by reducing the activity of the enzyme Acc1, thus decreasing acyl chain length and restoring membrane fluidity.




Lipotype Lipidomics technology can characterize whole-cell lipid composition to support the study of membrane biophysics, or focus on giant plasma membrane vesicles or nano plasma membrane vesicles. A combination of biophysical, genetic, functional, and lipidomic analyses can elucidate the relationships between membrane lipid composition, fluidity, and biological function. This approach may help resolve outstanding questions about disease pathology and/or the evolutionary role of specific lipids, for example, the role of phosphatidylinositol in the retina metabolism, and lipid metabolism genes.
Do our methods apply to your samples?
Let’s figure that out!

Lipotype products are provided for Research Use Only. They are not intended for clinical diagnostic purposes and must not be used to inform medical treatment decisions. The content of this article is for scientific and educational purposes only and should not be considered medical advice.