CANCER, or the uncontrolled growth of cells at the expense of the host, is estimated to be the second-leading cause of death worldwide. The most common forms of cancer include lung cancer, breast cancer, prostate cancer, colorectal cancer, and ovarian cancer, but the disease can develop in almost any organ in the human body. Disease progression occurs through a process called metastasis, in which cancer cells move from the organ of origin to a second site in the body.
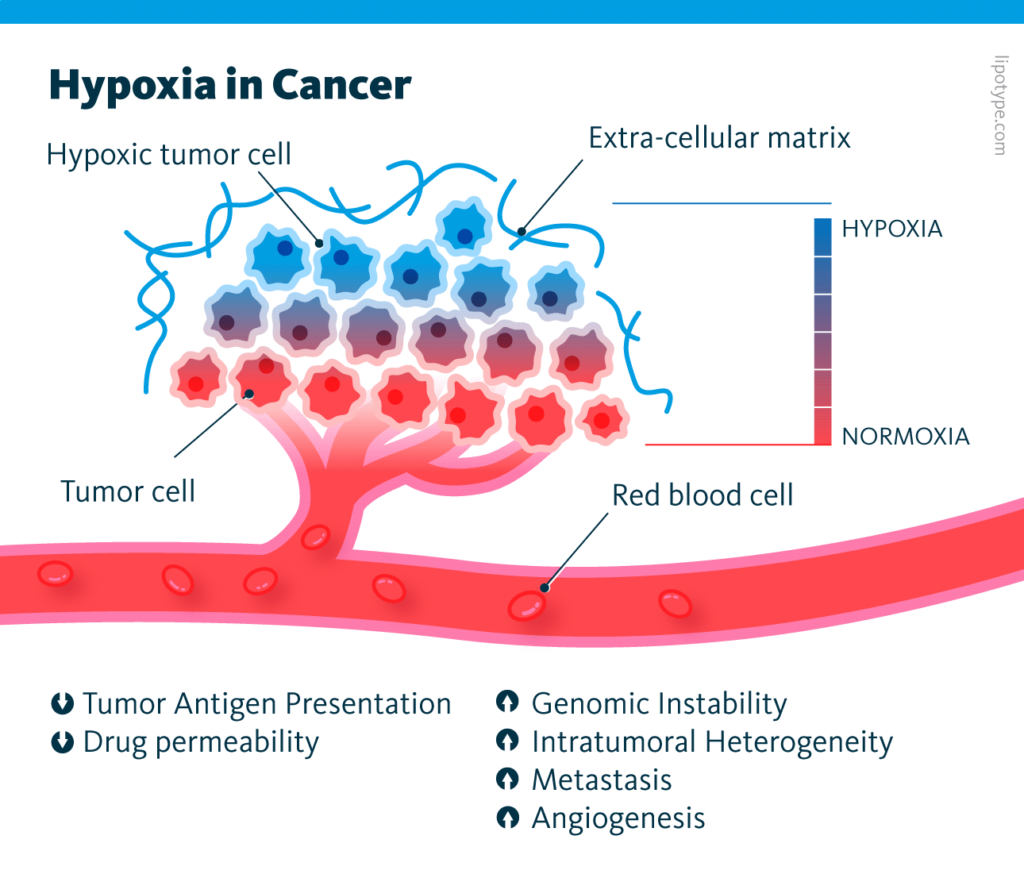
Though cancer is a heterogeneous disease, tumor microenvironments across organs share characteristic features. One of these features is hypoxia, or low oxygen tension.
As tumors grow, they quickly outgrow their blood supply. This leaves parts of the tumor tissue with regions where the oxygen concentration is lower than in healthy tissue. The resulting hypoxic microenvironment is associated with some hallmarks of cancer, including angiogenesis, immunosuppression, and metastasis. Interestingly enough, prostaglandins play an important role in many of these cancer-related processes in various cancers.
Hypoxia is considered a therapeutic target, but the molecular mechanisms underlying the role of hypoxia in cancer development and progression have not been fully described. A recent study reported by researchers at The University of Hong Kong seeks to identify and characterize the genes that enable cancer cells to survive in and adapt to hypoxia.
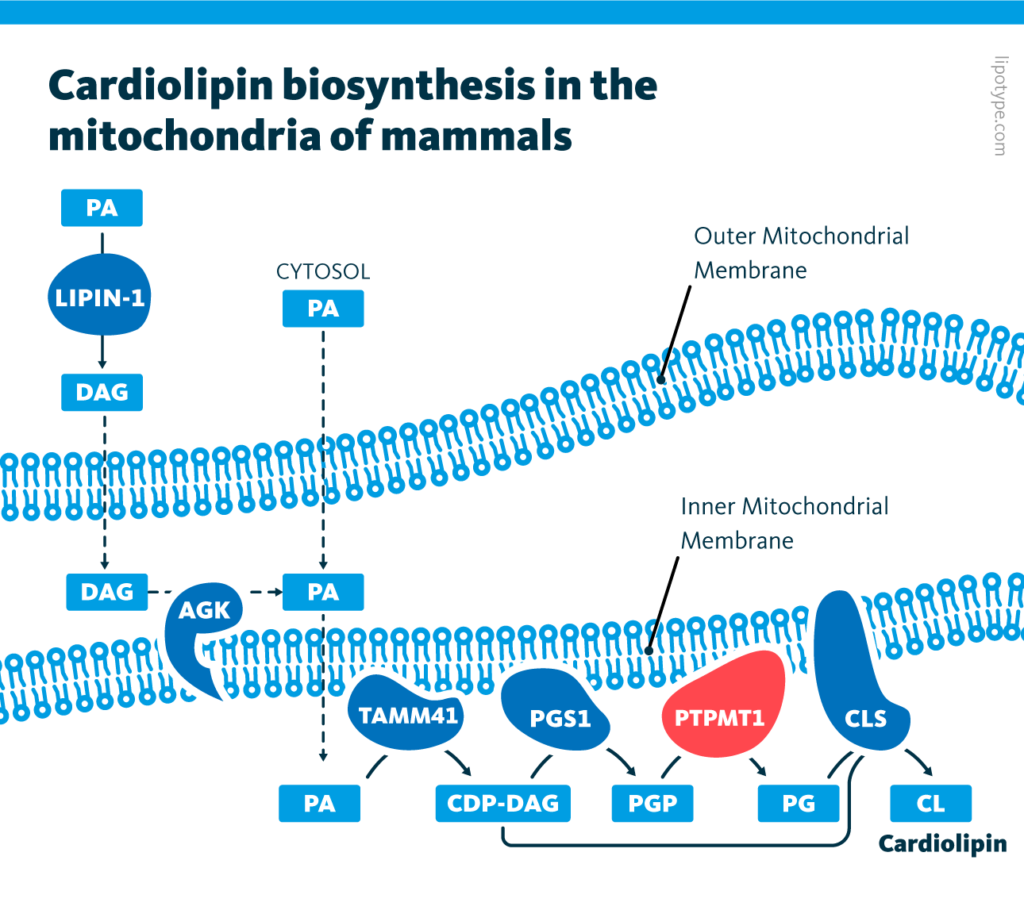
Cardiolipin biosynthesis in the mitochondria of mammals: The biochemical pathways of cardiolipin synthesis in the mitochondria of mammalian cells. Two sources of phosphatidates (PA) are shown. The enzyme PTPMT1 is highlighted.
The researchers created a CRISPR-Cas9 knockout library in hepatocellular carcinoma cells, a type of liver cancer that is often hypoxic. Following a hypoxia-driven negative selection process, this library revealed that the protein “protein-tyrosine phosphatase mitochondrial 1” (PTPMT1) enables cells to adapt to hypoxic conditions. Specifically, PTPMT1 knockout cells exhibited slower growth, lower viability, and less aggressive phenotypes both in vitro and in vivo – and hypoxia enhanced these changes.
PTPMT1 is an enzyme that contributes to biosynthesis of cardiolipin, a type of diphosphatidylglycerol lipid found in the membrane of mitochondria – or, the powerhouses of the cell. In PTPMT1 knockout cells, mitochondria had altered morphology and generated less energy. According to a lipid profile of hypoxic knockout cells, these changes in mitochondrial function were accompanied by a reduction in cardiolipin levels.
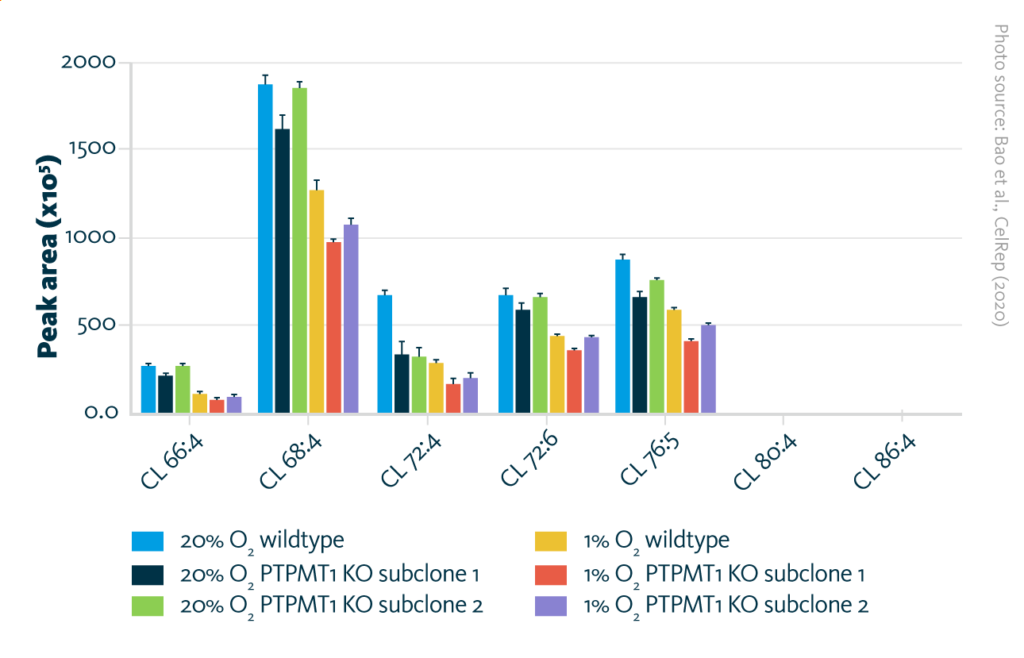
Cardiolipin composition under normoxia and hypoxia: Absolute levels of different cardiolipin species in PTPMT1 knockout subclones and wildtype after culturing in 20% oxygen (normoxia) or 1% oxygen (hypoxia).
Bao et al., Cell Rep (2020), doi: 10.1016/j.celrep.2020.108676
In multiple hepatocellular carcinoma cell lines and cell lines derived from ovarian, colorectal, breast, pancreatic, and prostate cancers, the PTPMT1 inhibitor alexidine dihydrochloride recreated the phenotypes observed in PTPMT1 knockout cells. Specifically, under hypoxic conditions, the inhibitor reduced cell growth, mitochondrial energy generation, and cardiolipin levels; increased apoptosis; and altered the cellular lipid profile and mitochondrial morphology. Interestingly enough, changes in lipid profiles are also involved in another type of programmed cell death – ferroptosis.
Moreover, the study showed that PTPMT1 is overexpressed in primary hepatocellular carcinoma biopsies from a mouse model. This result was supported by data from The Cancer Genome Atlas, which revealed that overexpression of PTPMT1 correlates with worse patient prognosis.
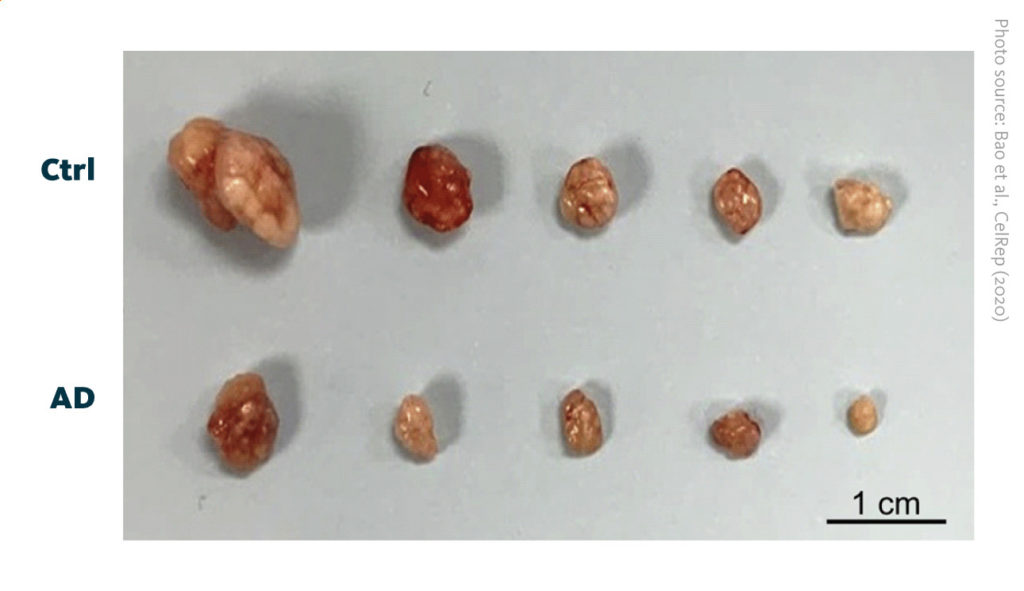
Suppressed tumor growth of hepatocellular carcinoma after disruption of cardiolipin synthesis pathway: Images of tumors from control (Ctrl) and alexidine dihydrochloride treated (AD) mice. AD tumors are smaller than Ctrl tumors.
Bao et al., Cell Rep (2020), doi: 10.1016/j.celrep.2020.108676
Overall, this study identifies and characterizes a key role for PTPMT1 in the adaptation of cancer cells to hypoxic conditions. Through overexpression of PTPMT1 and subsequent synthesis of cardiolipin, cancer cells recover their mitochondrial function and survive hypoxia – this is similar to how cardiolipin synthesis and remodeling is impacted during mtUPR.
Hypoxia is a common feature of cancer, but the specifics of how cells adapt to low oxygen conditions have not been fully characterized. Future work characterizing other genes identified in the recently reported CRISPR-Cas9 knockout library at the protein, lipid, organelle, cell, and animal levels may facilitate a more complete understanding of the role of hypoxia in cancer and inform the development of future targeted therapies.


Lipotype Lipidomics technology provides an unbiased characterization of cellular lipid profiles. These data about lipid identity and concentration can reveal and confirm hypotheses about the molecular mechanisms driving physiological and pathological processes, including those involved in hypoxia, mitochondrial function, and cancer.
Need clarity on the process?
Ask us anything!

Lipotype products are provided for Research Use Only. They are not intended for clinical diagnostic purposes and must not be used to inform medical treatment decisions. The content of this article is for scientific and educational purposes only and should not be considered medical advice.