by Dr. Mariia Kuzina
FERROPTOSIS is a regulated form of cell death that is associated with the accumulation of lipid hydroperoxides and reactive oxygen species that break down fats in cellular membranes. This process differs from other forms of programmed cell death, as the genes that regulate ferroptosis have no role in regulating any other form of cell death, offering a potential avenue for optimizing cancer treatment.
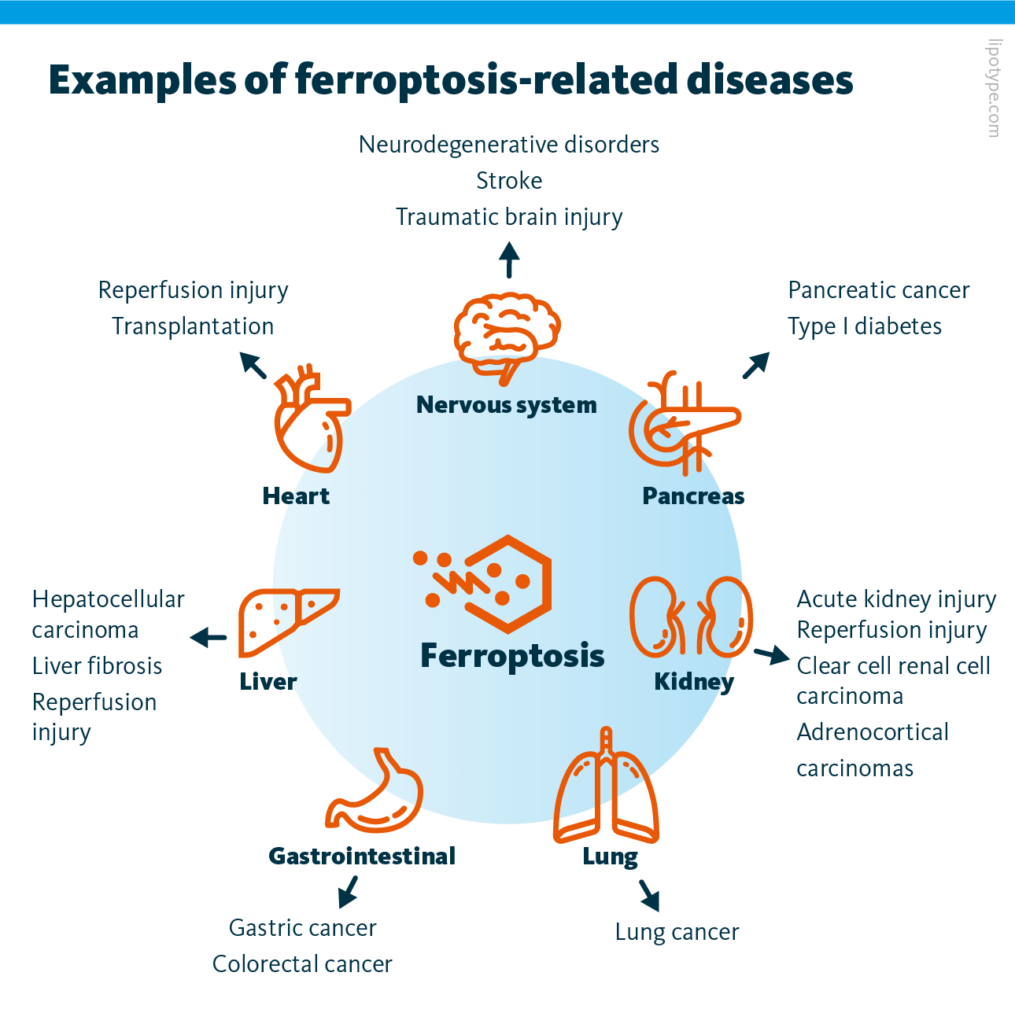
Ferroptosis plays a role in various diseases, including neurodegeneration, cardiovascular diseases, kidney disorders, and various types of cancer. To optimize targeted ferroptosis induction in cancer cells, researchers have been focused on inhibiting cellular antioxidant pathways that suppress ferroptosis, for example, regulating the activity of the enzyme GPX4 (glutathione peroxidase 4) inhibiting lipid metabolism.
Researchers have also pursued a complementary strategy for ferroptosis induction by enhancing the production of lipid hydroperoxides, which can spread through a free radical-mediated chain reaction to increase cell death. This approach has shown promise in sensitizing cancer cells to ferroptosis-triggering agents, such as GPX4 inhibitors, for example by increasing polyunsaturated lipids in cancer cells, which have been found to enhance their vulnerability to ferroptosis.
The recent study of Beatty et al. reports that linolenic fatty acids with conjugated double bonds, presented in many plant species, can induce ferroptosis in diverse cancer cells. Researchers found that αESA triggered cell death as a single agent and this death was suppressed by fer-1, the iron chelator deferoxamine, and by the lipophilic antioxidant vitamin E. Cell death by ferroptosis was associated with an increase in lipid peroxidation products that could be otherwise suppressed by fer-1.
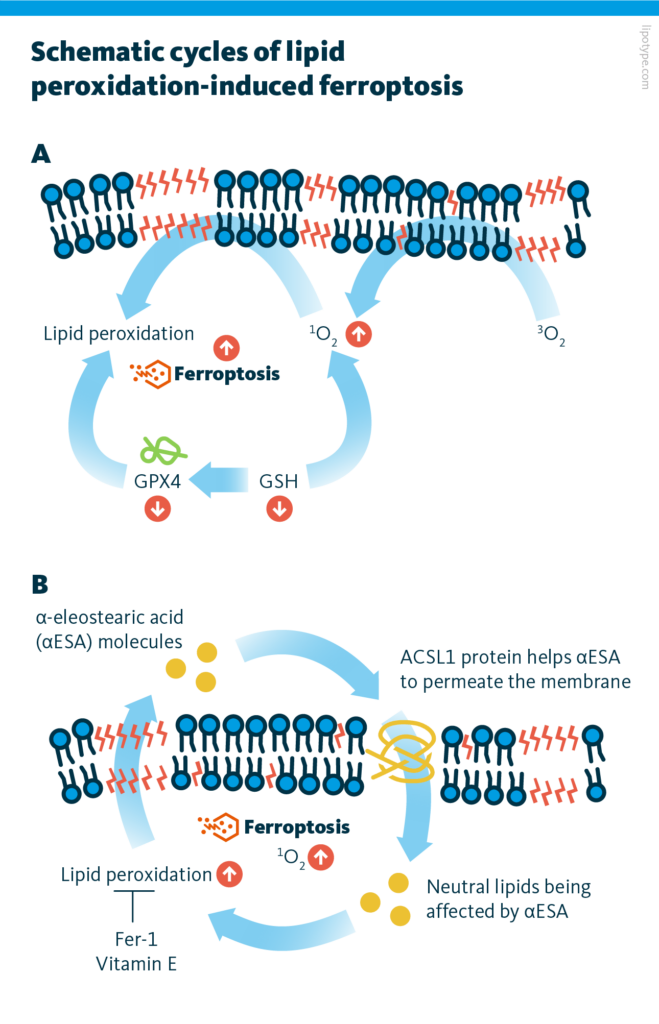
Schematic cycle of lipid oxidation-induced ferroptosis. A The role of GPX4 in ferroptosis induction. B The role of ACSL1 and αESA in lipid peroxidation and ferroptosis induction. Active components taking part in the ferroptosis: GPX4 inhibitors, αESA linolenic fatty acids affecting neutral lipids and increasing polyunsaturated lipids in cancer cells, the formation of lipid hydroperoxides. However, the ferroptosis can be suppressed by fer-1 (iron chelator deferoxamine) and the lipid antioxidant vitamin E.
Beatty et al., Nature Communications (2021) 12:2244, 10.1038/s41467-021-22471-y
This study examined how cell toxicity depends on various structural features of the αESA and screened related polyunsaturated fatty acids for death induction. Results showed that αESA with conjugated double bonds at carbons 9, 11, and 13 induced cell death. Conjugated linoleic acid was a poor inducer of cell death, suggesting the third conjugated double bond is required for ferroptosis activity.
Jacaric and catalpic acids were consistently the most toxic to the panel, followed by α-calendric acid, αESA, and βESA. Punicic acid and arachidonic acid exhibited substantially less anti-proliferative activity and the remaining control fatty acid were largely inactive. These results support a unique ferroptosis-inducing activity of conjugated fatty acids.
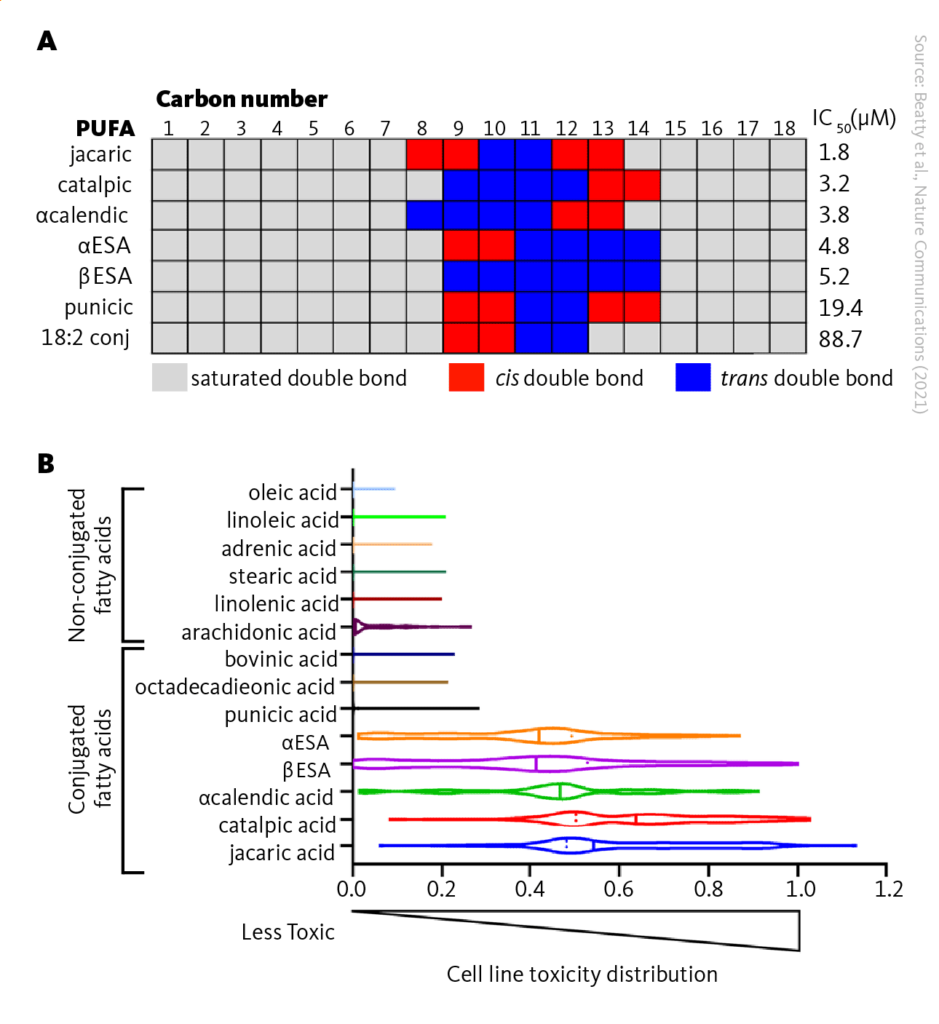
The toxicity of conjugated linolenic fatty acids for cancer cells. A The correlation of the structure and effectiveness of 18-carbon conjugated polyunsaturated fatty acids against cancer cell line after 72 hours of treatment. B Toxicity of different fatty acids on a mix of 100 human cancer cell lines and measured cell viability after 48 hours. The results are shown in a violin plot with a toxicity value of 1 representing complete cell loss and 0 indicating normal viability; IC50, half maximal inhibitory concentration.
Beatty et al., Nature Communications (2021) 12:2244, 10.1038/s41467-021-22471-y
The researchers further used lipidomics to investigate the role of ACSL1 (a protein involved in lipid metabolism) in the production of different types of lipids. The researchers found that the abundances of diacylglycerols (DAGs) and triacylglycerols (TAGs) were significantly reduced in ACSL1-deficient cells compared to control cells. Additionally, they treated cells with αESA, and specifically evaluated lipids with 18:3 acyl chains. It was found that their mole fraction was significantly decreased in ACSL1-deficient cells compared to control cells. This suggests that the resistance of ACSL1-deficient cells to αESA may be related to decreased αESA incorporation.
The researchers also observed changes in other lipid subgroups, including cholesterol esters and phosphatidylcholine, which may be due to weak utilization of αESA by other ACSL isoforms. Overall, lipidomics analysis was applied to identify changes in lipid metabolism in response to ACSL1 modulation and to gain insight into the potential mechanisms underlying these changes.
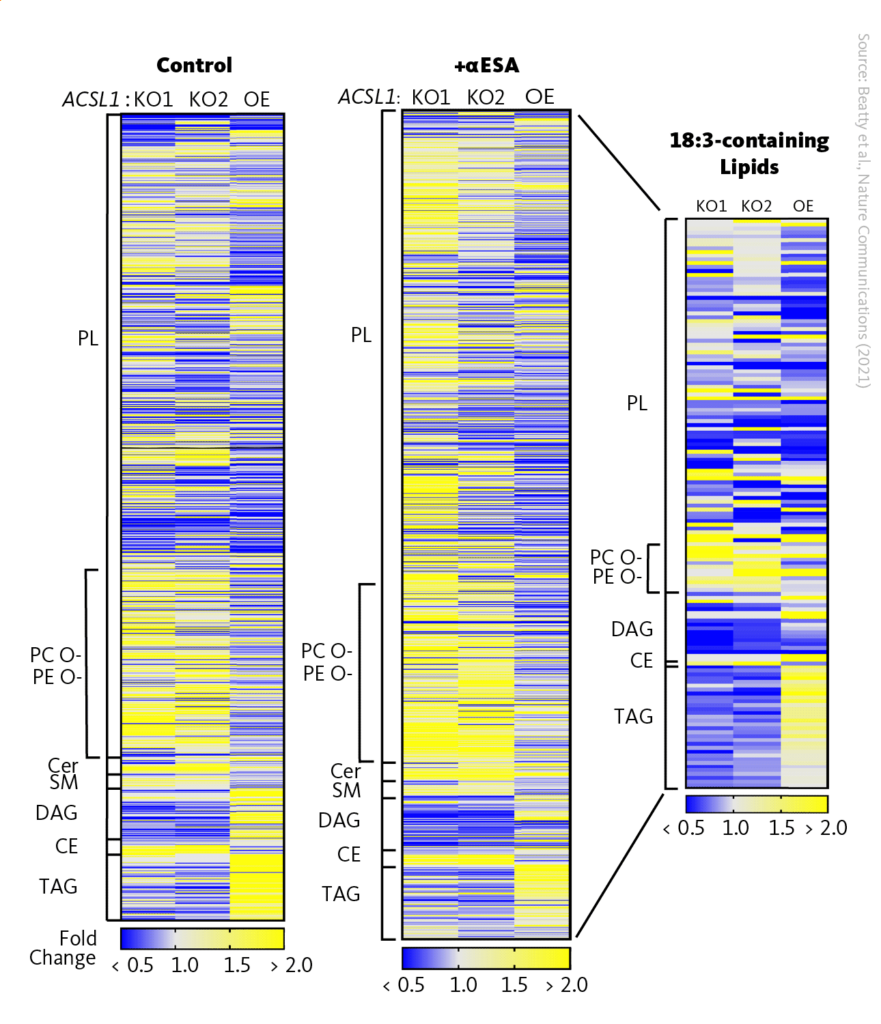
ACSL1 drives incorporation of αESA into neutral lipids. Heatmaps representing relative lipid abundance in ACSL1-deficient or overexpressing cells as fold-change in mole percent compared to control cells after 16 h treatment with αESA (right and middle) or in untreated cells (left). The rightmost heatmap shows only the subset of lipids with 18:3 acyl chains. Yellow indicates an increase in relative abundance and blue shows a decrease in relative abundance. PL, phospholipids; CE, cholesterol esters; Cer, ceramide; DAG, diacylglycerol; PC O-, ether-linked phosphatidylcholine; PE O-, ether-linked phosphatidylethanolamine; SM, sphingomyelin; TAG, triacylglycerol.
Beatty et al., Nature Communications (2021) 12:2244, 10.1038/s41467-021-22471-y
By using liquid chromatography-mass spectrometry, the authors measured 141 lipid species in different categories of cell membranes. They found that cells treated with αESA acid had more oxidized lipids compared to untreated cells. Previous studies have shown that various oxidized lipids increase in cells undergoing ferroptosis, but the authors focused on two specific types of lipids, arachidonic or adrenic acids, that were reported to have a significant role in this process.
They found that 80% of the oxidized lipids that changed significantly in the αESA-treated cells were increased. This result supports their observation of increased lipid peroxidation products in these cells. They also found that when ACSL1 was silenced, 24% of the increased oxygenated lipids in αESA-treated cells were decreased.
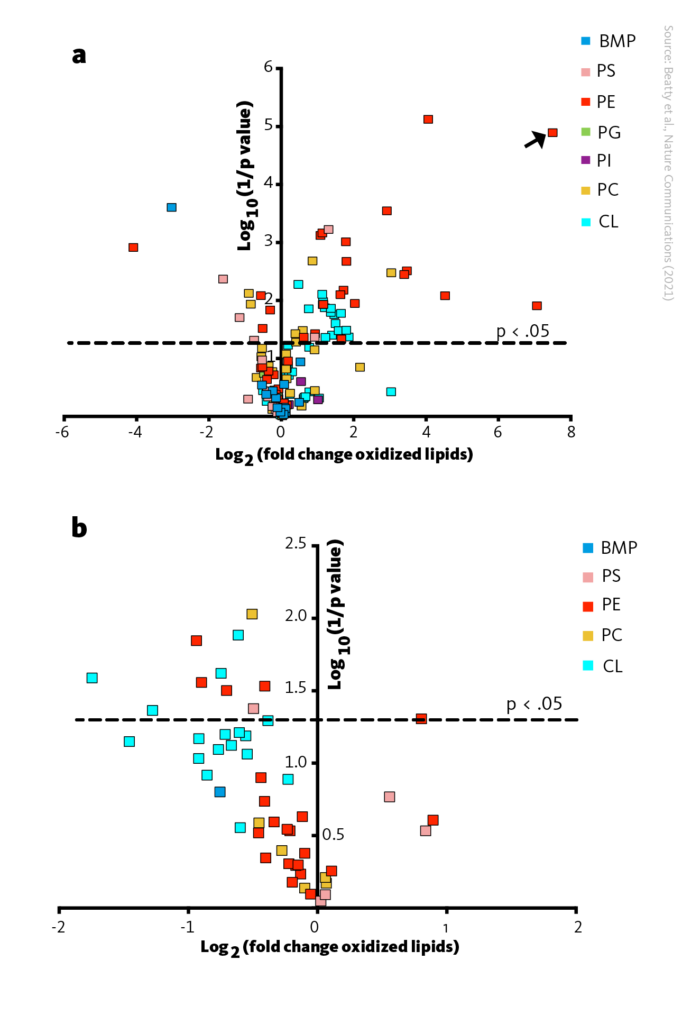
ACSL1 promotes lipid peroxidation by αESA. A Volcano plot visualizing log2(fold change) and significance (log10(1/p)) for oxidized phospholipids in cells incubated with αESA compared to control. The arrow points out a lipid species which was only detected after αESA treatment. B Volcano plot visualizing log2(fold change) versus log10(1/p-value) for oxidized phospholipids in cells transfected with a pool of ACSL1 siRNA and incubated with αESA compared to control. The set is limited to the 51 oxidized phospholipid species that were significantly changed by αESA treatment as shown in A. BMP, bis(monoacylglycero)phosphate; PS, phosphatidylserine; PE, phosphatidylethanolamine; PG, phosphatidylglycerol; PI, phosphatidylinositol; PC,phosphatidylcholine; CL, cardiolipin.
Beatty et al., Nature Communications (2021) 12:2244, 10.1038/s41467-021-22471-y
This study compares a typical GPX4 inhibitor-promoted ferroptosis to the αESA-promoted ferroptosis, focusing on the enhancement of hydroperoxide production by αESA. The authors confirmed their observations by adjusting the diet of the mice with the xenograft model of late-stage breast cancer. The mice were fed with tung oil, which contains αESA, and they showed slowed down growth and spreading of tumors. Finally, the tumors showed changes in gene expression consistent with ferroptosis.
Both cell culture and animal model experiments suggest that combining αESA with GPX4 inhibitors may increase its effectiveness as an anti-tumor treatment. The results provide insight into ferroptosis regulation and offer a potential therapeutic approach to cancer treatments.



Lipotype Lipidomics technology can provide a detailed analysis of lipid profiles and help researchers understand the role of lipids in ferroptosis and apoptosis, which can ultimately lead to the development of new treatments for diseases associated with these forms of cell death.
Do our methods apply to your samples?
Let’s figure that out!

Lipotype products are provided for Research Use Only. They are not intended for clinical diagnostic purposes and must not be used to inform medical treatment decisions. The content of this article is for scientific and educational purposes only and should not be considered medical advice.